COVID-19 Research
Principal Investigators

Prof. Matthias Matter
Leitender Arzt und Fachbereichsleiter Molekularpathologie
Pathologie
Tel. +41 61 328 64 71

Prof. Alexandar Tzankov
Leitender Arzt und Fachbereichsleiter Histopathologie und Autopsie
Pathologie
Tel. +41 61 328 68 80
Group Members



Anna Stalder
Postdoc
Pathologie
Our Science
SARS-CoV-2 interactions with human cells, tissues and organs in COVID-19 patients
A rapidly evolving sweeping pandemic caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) leading to coronavirus disease 2019 (COVID-19) has dominated the first half of 2020 and continues to do so. Cases of COVID-19 were first described in late 2019, when a series of previously unidentified pneumonia-related deaths emerged in Hubei province, China. COVID-19, subsequently spread to almost all countries with more than half a million confirmed deaths worldwide (https://www.worldometers.info/coronavirus/).
Although virus’ origin, cellular entry and epidemiology have rapidly been clarified, in situ observations of the actual viral interactions within human organs and tissues in patients suffering from COVID-19 have for a long time been addressed at the level of case reports or small series of ≤4 cases. Indeed, by the end of April 2020 when 150.000 patients had already died of COVID-19, only 16 autopsy cases had been reported in the peer-reviewed literature, with nine publications presenting limited autopsies, assessment of postmortem core-needle or incisional collection of tissue. In the absence of reliable data regarding the degree of SARS-CoV-2 infectivity in dead individuals, various authorities discouraged the conduction of autopsies. This, combined with the ill-adjusted attitudes of pathologists, clinicians and societies towards autopsies, locked down scientific activities to elucidate the actual underlying mechanisms of COVID-19. This seems incomprehensible, given the fundamental and time-proven role of autopsies in re-emerging, emerging or unknown diseases. Only after 170.000 reported COVID-19 deaths and four months of pandemic, the first autopsy series of >10 patients (n=21), i.e. the Basel autopsy cohort, was published (Menter et al.).
Autopsy findings from this cohort provided initial broad evidence of a rather characteristic morphologic pattern of lung changes in COVID-19 with deep-red, slightly nodular, hyperemic and very heavy lungs with prominent diffuse alveolar damage, and, most importantly, microvascular thrombosis and capillary congestion (Figure 1).
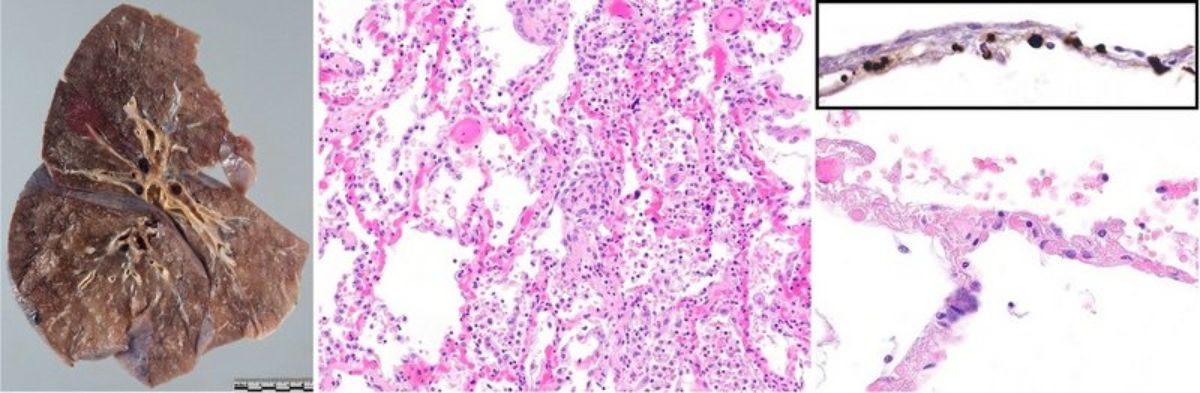
Figure 1. Left: Gross appearance of a COVID-19 lung. Middle: Diffuse alveolar damage with hyaline membranes in a patient with lethal COVID-19 with prominent capillary stasis. Right: Upon higher magnification, thrombotic occlusion can be observed in alveolar capillaries. Inset: Immunohistochemical staining for fibrin reveals occlusion of alveolar capillaries by microthrombi.
It was the collected material from Basel to allow uncovering of rater unique and distinctive vascular features consisting of severe endothelial injury, associated with intracellular SARS-CoV-2, widespread thrombosis, microangiopathy and new vessel growth—predominantly by the mechanism of intussusceptive angiogenesis (Figure 2), allowing to conclude that COVID-19 is an angiocentric clinical syndrome [Ackermann et al.].
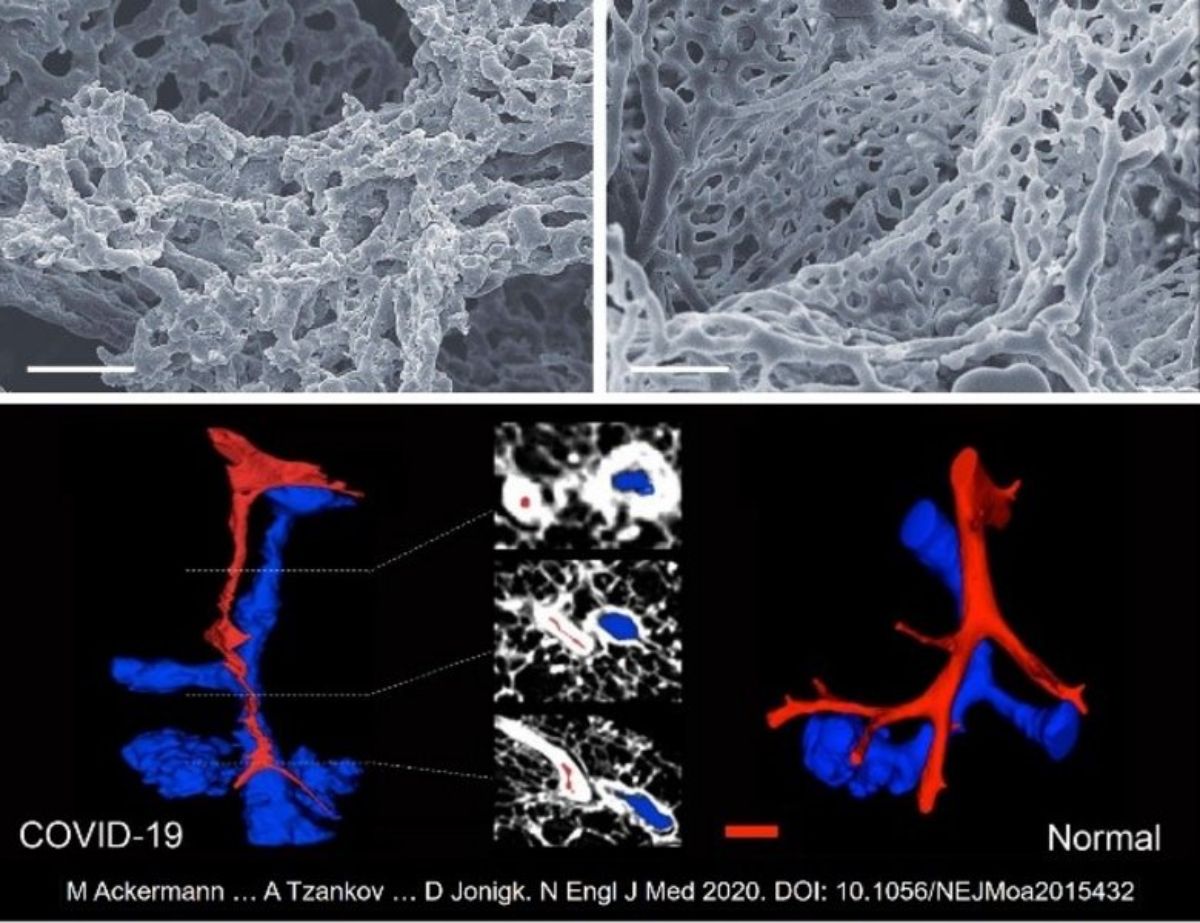
Figure 2. Upper row: Scanning electron micrographs of microvascular corrosion casts from the thin-walled alveolar plexus of an injured by COVID-19- (left) and a healthy lung (right).
Lower row: (Sub)total occlusion of the arteries (red) in COVID-19-lungs (left), as compared to uninfected controls (right). (μCT 3D reconstruction)
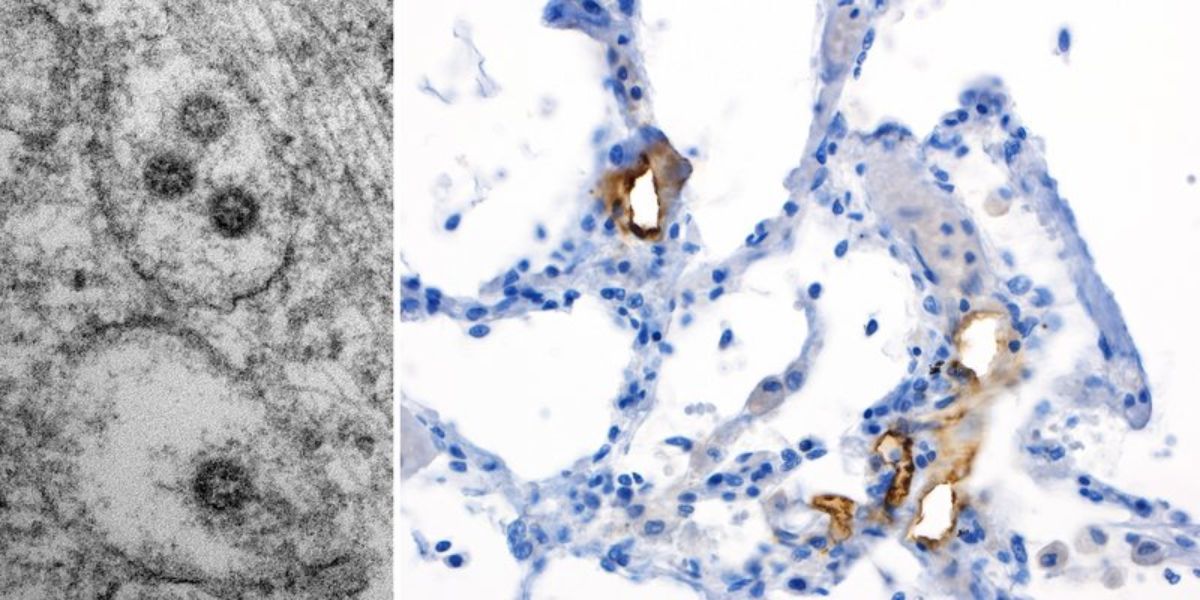
Figure 3. Left: Electron microscopic images of virus structures of characteristic size (75-120nm) within membranous intracellular structures of the trans-Golgi apparatus/endoplasmic reticulum with characteristic nucleocapsid structures on the inner side (~20nm measuring dark dots), and merely perceptible surrounding projections. Right: Immunohistochemical staining of a lung specimen for SARS-CoV-2 Spike protein with the clone 007 from Sino Biological on an autopsy case from our published cohort showing presence of spiked viruses mainly in the endothelial compartment.
Indeed, not only are virus-like particles detectable in endothelial cells, but applying anti-SARS-CoV-2-spike-protein antibodies, one will detect highly preferential presence of virus antigens in the endothelial, but not in the epithelial compartment of the alveolar units (Figure 3), arguing in favor of the above hypothesis.
Based on the above mentioned, the research proposal of the COVID-19 research group at or institute was selected by the Fast Track Call for Acute Global Health Challenges of the Botnar Research Centre for Child Health (BRCCH) and granted 1.3 Million CHF for the next 2.5 years. In an interdisciplinary approach with researchers from pathology, immunology, internal medicine, intensive care and neurology, knowledge of the pathogenesis of COVID-19 with the final goal to affect future more tailored medical interventions for this disease is being generated now.
The main research topics are:
- Tissue and cellular tropism of SARS-CoV-2 and in situ imaging of coronavirus reservoirs
- Immunohistochemical analysis of COVID-19 tissues with respect to virus proteins, inflammatory cellular composition and alteration of angiotensin-cascade compounds
- RNA expression analysis with respect to SARS-CoV-2 and gene expression of angiogenesis-, tissue remodeling- and immunity/inflammation genes
- High parameter tissue imaging of coronavirus pathology (CODEX)
Selected Publications
- Tzankov A, Bhattacharyya S, Kotlo K, Tobacman JK. Increase in Chondroitin Sulfate and Decline in Arylsulfatase B May Contribute to Pathophysiology of COVID-19 Respiratory Failure. Pathobiology. 2021 Nov 17:1-11.
- Yekelchyk M, Madan E, Wilhelm J, et al. Flower lose, a cell fitness marker, predicts COVID-19 prognosis. EMBO Mol Med. 2021 Nov 8;13(11):e13714.
- Nakayama T, Lee IT, Jiang S, et al. Determinants of SARS-CoV-2 entry and replication in airway mucosal tissue and susceptibility in smokers. Cell Rep Med. 2021 Oct 19;2(10):100421.
- Hozayen SM, Zychowski D, Benson S, et al. Outpatient and inpatient anticoagulation therapy and the risk for hospital admission and death among COVID-19 patients. EClinicalMedicine. 2021 Nov;41:101139. doi: 10.1016/j.eclinm.2021.101139.
- Menter T, Cueni N, Gebhard EC, et al. Case Report: Co-occurrence of Myocarditis and Thrombotic Microangiopathy Limited to the Heart in a COVID-19 Patient. Front Cardiovasc Med. 2021 Jul 29.
- Obermayer A, Jakob LM, Haslbauer JD, et al. Neutrophil Extracellular Traps in Fatal COVID-19-Associated Lung Injury. Dis Markers. 2021 Jul 30.
- Sahanic S, Löffler-Ragg J, Tymoszuk P, et al. The Role of Innate Immunity and Bioactive Lipid Mediators in COVID-19 and Influenza. Front Physiol. 2021 Jul 22;12:688946.
- Menter T, Tzankov A, Bruder E. SARS-CoV-2/COVID-19-Auswirkungen auf die Plazenta [Impact of SARS-CoV-2/COVID-19 on the placenta]. Pathologe. 2021 Jun 11:1–7.
- Wu CT, Lidsky PV, Xiao Y, et al. SARS-CoV-2 infects human pancreatic β cells and elicits β cell impairment. Cell Metab. 2021 Aug 3;33(8):1565-1576.
- Boor P, Eichhorn P, Hartmann A, et al. Praktische Aspekte von COVID-19-Obduktionen [Practical aspects of COVID-19 autopsies]. Pathologe. 2021 Mar;42(2):197-207.
- Bösmüller H, Matter M, Fend F, et al. The pulmonary pathology of COVID-19. Virchows Arch. 2021 Feb 19:1–14.
- Haslbauer JD, Matter MS, Stalder AK, et al. Reaktionsmuster der lokoregionären Lymphknoten im Abflussgebiet von COVID-19-Lungen [Histomorphological patterns of regional lymph nodes in COVID-19 lungs]. Pathologe. 2021 Mar;42(2):188-196.
- Zellweger NM, Huber J, Tsakiris DA, et al. Haemophagocytic lymphohistiocytosis and liver failure-induced massive hyperferritinaemia in a male COVID-19 patient. Swiss Med Wkly. 2021 Jan 15;151:w20420.
- Haslbauer JD, Perrina V, Matter M, et al. Retrospective Post-mortem SARS-CoV-2 RT-PCR of Autopsies with COVID-19-Suggestive Pathology Supports the Absence of Lethal Community Spread in Basel, Switzerland, before February 2020. Pathobiology. 2021;88(1):95-105. doi: 10.1159/000512563.
- Menter T, Tzankov A. Investigations of Pathologists as a Key to Understanding Coronavirus Disease 2019. Pathobiology. 2021;88(1):11-14.
- Reinhold A, Tzankov A, Matter M, et al. Ocular pathology and occasionally detectable intraocular SARS-CoV-2 RNA in five fatal COVID-19 cases. Ophthalmic Res. 2021 Jan 20.
- Glatzel M, Hagel C, Matschke J, et al. Neuropathology associated with SARS-CoV-2 infection. Lancet. 2021 Jan 23;397(10271):276.
- Cribiù FM, Erra R, Pugni L, et al. Severe SARS-CoV-2 placenta infection can impact neonatal outcome in the absence of vertical transmission. J Clin Invest. 2021 Jan 26:145427.
- Fuchs V, Kutza M, Wischnewski S, et al. Presence of SARS-CoV-2 Transcripts in the Choroid Plexus of MS and Non-MS Patients With COVID-19. Neurol Neuroimmunol Neuroinflamm. 2021 Jan 27;8(2):e957.
- Lee IT, Nakayama T, Wu CT, et al. ACE2 localizes to the respiratory cilia and is not increased by ACE inhibitors or ARBs. Nature Communications. 2020 Oct 28;11(1):5453.
- Nienhold R, Ciani Y, Koelzer VH, et al. Two disctinct immunopathological profiles in autopsy lungs of COVID-19. Nature Communications. 2020 Oct 8;11(1):5086.
- Frank S. Catch me if you can: SARS-CoV-2 detection in brains of deceased patients with COVID-19. Lancet Neurol. 2020 Oct 5;19(11):883–4.
- Hopfer H, Herzig MC, Gosert R, et al. Hunting coronavirus by transmission electron microscopy - a guide to SARS-CoV-2-associated ultrastructural pathology in COVID-19 tissues. Histopathology. 2020 Sep 27:10.1111/his.14264.
- Menter T, Mertz KD, Jiang S, et al. Placental Pathology Findings during and after SARS-CoV-2 Infection: Features of Villitis and Malperfusion. Pathobiology. 2020 Sep 18:1-9.
- Ackermann M, Verleden SE, Kuehnel M, et al. Pulmonary vascular endothelialitis, thrombosis, and angiogenesis in Covid-19. The New England Journal of Medicine. 2020 383:120-128
- Deigendesch N, Sironi L, Kutza M, et al. Correlates of critical illness-related encephalopathy predominate postmortem COVID-19 neuropathology. Acta Neuropathologica. 2020 Aug 26;1-4.
- Eckermann M, Frohn J, Reichardt M, et al. 3D virtual pathohistology of lung tissue from Covid-19 patients based on phase contrast X-ray tomography. eLife. 2020 Aug 20;9:e60408. doi: 10.7554/eLife.60408.
- Jamiolkowski D, Mühleisen B, Müller S, et al. SARS-CoV-2 PCR testing of skin for COVID-19 diagnostics: a case report; The Lancet. 2020 396(10251):598-599
- Löffler KU, Reinhold A, Herwig-Carl MC, et al. Ocular post-mortem findings in patients having died from COVID-19. Ophthalmologe. 2020 117:648-651
- Menter T, Haslbauer JD, Nienhold R, et al. Post-mortem examination of COVID19 patients reveals diffuse alveolar damage with severe capillary congestion and variegated findings of lungs and other organs suggesting vascular dysfunction. Histopathology. 2020 77:198-209
- Mohanty SK, Satapathy A, Naidu MM, et al. Severe Acute Respiratory Syndrome Coronavirus-2 (SARS-CoV-2) and Coronavirus Disease 19 (COVID-19) – anatomic pathology perspective on current knowledge; Diagnostic Pathology. 2020 15:103.
- Tzankov A, Jonigk D. Unlocking the lockdown of science and demystifying COVID-19: how autopsies contribute to our understanding of a deadly pandemic. Virchows Archiv. 2020 Sep;477(3):331-333.
Awards and Grants
Collaborations
- Institute of Pathology, Cantonal Hospital Baselland, Liestal, Switzerland
- Intensive Care Unit and Division of Internal Medicine, University Hospital Basel, University of Basel, Basel
- Institute of Pathology, Hannover Medical School, and the German Center for Lung Research, Biomedical Research in Endstage and Obstructive Lung Disease Hannover, Germany
- Departments of Neurosurgery and Radiology, University Hospital Basel, University of Basel, Basel, Switzerland
- Nolan Lab, Stanford University, Stanford, CA, USA
